Data table 4 liquid measurements10/31/2023 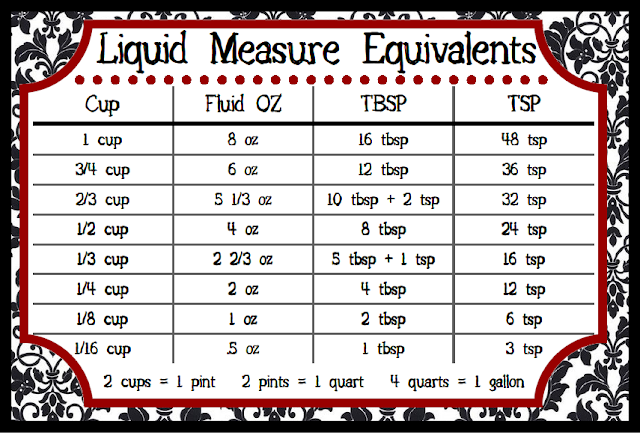
When making measurements, both precision and accuracy are extremely important. If you are not with in an error range of +/- 5% then try again.Determine the % error as described in the previous section and record on Table 3.If you accurately dispensed 3.6 mL, the weight difference should be very close to 3.6 g. Determine the weight of the water and record.Record this value in your table as “weight of container and water.” Dispense the 3.6 mL into the weighed beaker and determine its mass (g).To get the last bit out of the pipet, quickly dial one way then the other. Dispense the water into the weighed beaker dialing in the opposite direction. 
Use the dial to draw 3.7 mL of deionized water into the pipet from a 50 mL graduated cylinder. Do not shove the pipet way up into the pump. Obtain a 5 mL serological pipet and a pipet pump.Write the weight of the container you previously attained. You already weighed this container in the previous exercise. Dry the 50 ml beaker you used previously.If you are not with in an error range of +/- 5% then try again.∖ * 100 ∖ ]" id="MathJax-Element-6-Frame" role="presentation" style="position:relative " tabindex="0"> ∖ ] Determine the % error for each of your measurements as follows:.Because water has a density of 1g/mL, then the number g=mLs (50ml=50g) Record this number as “actual volume dispensed”. Determine the weight of the water and record as “weight of water only”. 
Weigh the beaker with the water and record on Table 3 under “weight of container and water.”. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
